What Reduces Battery Life
What Reduces Battery Life
Batteries have become the lifeblood of today’s machines, devices, and equipment. Batteries enable us to use them without an electrical connection.
But batteries, as we know, have limited capacity. Sooner we’ll have to plug our laptops, our phones, or replace our car batteries.
Batteries have a predetermined lifespan as indicated by the manufacturers’ specifications. However, some factors and actions can be too damaging to batteries that they reduce the battery’s life.
A battery can only last long if you know the ways to properly use and maintain it. Here are some of the factors that affect battery life:
Heat
We all know that it’s a bad practice to leave our phones or any other gadgets outside while it’s hot because it’s damaging. But what does heat do to batteries? How does heat affect the life of batteries of phones, laptops, cars, and a lot more?
Heat is a major battery killer because it forces the batteries to work harder. If they are exposed to extreme temperatures, they will exhibit abnormal behavior. It’s not because of a manufacturer’s defect but as a consequence of neglecting its specifications.
In some cases, when batteries are exposed to too much heat, they will simply not work. They will bubble, create sparks and flames, damage the device, or blow up.
But for most batteries, it’s a slow nearing death. As the temperature rises, electrode activities also increases. When this activity acclimates, adverse chemical reactions will take place such as corrosion, gassing, and passivation of electrodes. This will impact the self-discharge rate of batteries and will lead to a reduced lifespan. Once the damage has been done, the lifespan can’t be reverted to its normal capacity.
A lead-acid battery, for example, loses half of its life when exposed to 8°C (46°F) beyond its accepted range. VRLAs, the kind of battery used in submarines and big vehicles and power applications, have a lifespan of 10 years at a constant temperature of 25°C (77°F). But when it’s continuously exposed to temperatures higher than what’s acceptable, say 33°C (91°F), VRLAs will only live for five years. If kept at even higher temperatures the lifespan will reduce to 30 months.=
Nowadays, flooded lead-acid batteries are more reliable systems than their VRLA counterparts. Lead-acid ones can withstand high temperatures enabling them to have up to 20 years of lifespan. Of course, it has its disadvantages like the constant need for watering and good ventilation. But considering how it can ward off the damaging effects of heat, it’s worth it.
The good news is batteries are becoming more heat resistant. The Battery Council International Failure Mode Study has proven this. From just a heat tolerance of 7°C (44°F) in 2000, starter batteries have improved its heat resistance to 12°C (53°F) in 2010. Still, beyond that temperature, batteries are likely to decrease their lifespan by a year.
In other studies, starter batteries have lasted 34 months in 1962. By 2000, life expectancy rose to 41 months. Ten years after, BCI reported that batteries have an average lifespan of 55 months. Northern places have longer battery life because of their cooler climates than their Southern counterpart.
However, the battery’s ever-increasing lifespan must not eliminate the fact that heat resistance has limits. Although it went longer than ever. High heat, especially a repeated exposure, can still shorten its life and its damages are irreversible. The result will be just to deal with the short-live battery and replace them sooner than intended.
Monitoring systems can be deployed to check the health of batteries and their operating temperature. AKCP provides a battery health monitoring sensor that incorporated voltage, current and temperature readings into one sensor. In addition a temperature only, battery terminal temperature sensor can be used for simpler applications.
Battery Activity
Battery life depends on how it’s used. The activity of the user heavily impacts not just battery performance, but also its lifespan.
It’s a known fact that the more you use a battery, the faster you will drain it. How so? It has something to do with the discharge rate. Discharge cycles happen when the battery loses its capacity to provide power and needed to be recharged.
Batteries have a limited number of discharge or recharge cycles before the battery chemistry runs out. Cells will fail if the chemistry is exhausted. Soon the battery needs replacement.
If someone frequently uses the battery, the discharge rate is higher. A higher discharge rate means lower capacity. In other words, frequent use of battery lessens lifespan.
This activity is evident in phones, laptops, and most cars. Cranking the engines several times a day stresses starter batteries. But this is a bit different from the start-stop operation of a micro-hybrid. At red traffic lights, micro-hybrid turns off the internal combustion engine (ICE). When the traffic flows, it turns the ICE on. This results in about 2000 micro-cycles per year. Studies show the battery capacity dropped to 60% after two years of use. The solution is to use specialty AGM and other systems.
Cadex laboratory studies show a drop of 50% when the battery was exposed to 700 microcycles.
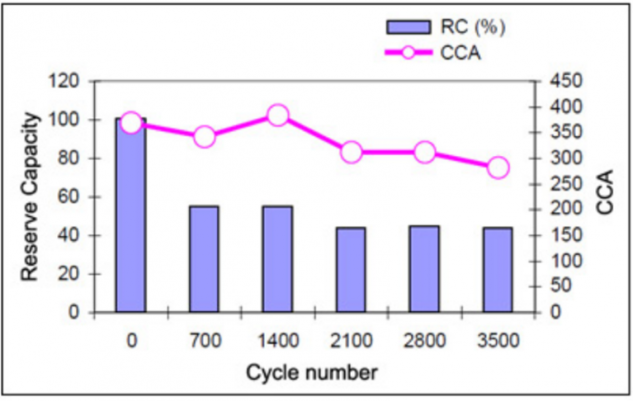
Frequent activity reduces battery lifespan but so is zero activity. If the battery goes long without use, charge carriers won’t produce any current, hence they will be consumed due to inactivity. Sooner, the battery can’t produce power altogether.
To prevent internal self-discharge, you must regularly use your batteries. Not using them equates to damaging them. But just be careful not to over-discharge them it also reduces the lifespan.
Conclusion
Battery lifetimes tend to fade over time. Some measures cannot be avoided like leaving the car on a hot summer road or leaving them in the cold. Cellphones can be discharged many times. And cars have a stop and go mechanisms that reduce their capacity. But knowing some of these ways can influence the actions of people who use the batteries. After all, battery life is not the only thing to be considered here. Some battery activities are extremely unsafe and hazardous to users. But the good news is that batteries of today are more resistant and it will get even better through the years as statistics show. It will potentially ward off some of its problems and we’ll soon have to deal with more efficient, better batteries.
Battery monitoring solutions can aid in assessing your battery performance, health and lifespan.
- About the Author
- Latest Posts

For over two decades at AKCP, I have been focused on a single mission: bringing complete visibility, security, and efficiency to the world’s critical infrastructure.
I believe that in the modern data center, AI is only as good as the data it receives. My goal is to ensure facilities have the precise sensor facts needed to control AI opinions, ultimately reducing PUE, releasing stranded capacity, and ensuring maximum uptime.

